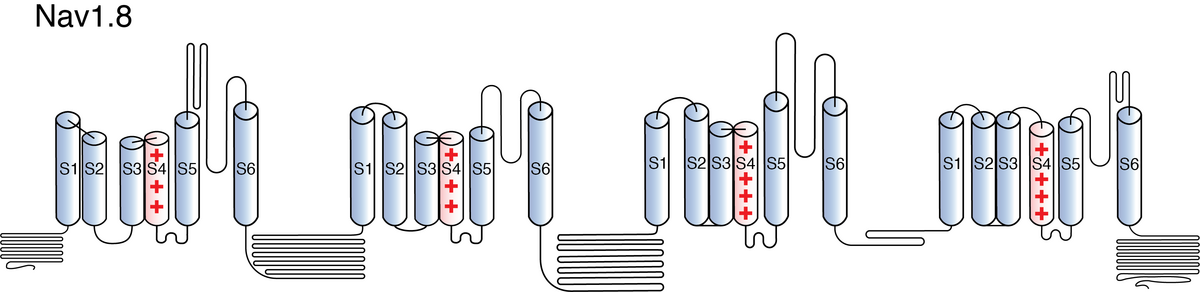
Nav1.8
Description: sodium channel, voltage-gated, type X, alpha subunit Gene: Scn10a Alias: nav1.8, scn10a, PN3, SNS, hPN3
Nav1.8 (also known as PN3; SNS; hPN3), encoded by the gene scn10a, is a sodium, voltage-gated, type 10, alpha subunit channel. Nav1.8 is predominantly expressed in the PNS. It is involved in the action potential initiation and nociception. Mutations to the channel are often the cause of chronic pain and inflammation disorders.
scn10a is the coding gene for Nav1.8. In humans, it is located on chromosome 3 (3p22.2) and is made of 28 exons, 27 of which are coding and exon 1 being non-coding. scn10a is found on the same gene cluster as scn5a (Nav1.5) and scn11a (Nav1.9). All 3 genes contain an extra exon (17b) between exons 17 and 18, which corresponds to a section on the loop between domain II and domain III.
The scn10a transcript has a length of 6.5 kilobase [859]
| Species | NCBI accession | Length (nt) | |
|---|---|---|---|
| Human | NM_006514.4 | 6626 | |
| Mouse | NM_001205321.1 | 6416 | |
| Rat | NM_017247.2 | 6507 |
Human Nav1.8 is made up of 1,956 amino acids (aa) and has a molecular weight of 220 Kda.
Isoforms
Nav1.8 contains a number of glycosylation sites, indicating that the protein is likely glycosylated. However, the exact timing of glycosylation and its impacts on channel kinetics or expression have not been extensively studied [860].
Nav1.8 is subject to phosphorylation by various enzymes:
- PKA was shown to modulate Nav1.8 on phosphorylation sites, notably some located in the intracellular loop between DI-DII. Removal of these sites via mutagenesis lead to a right shift in activation and a slowing of current inactivation, indicating that phosphorylation enhances the channel’s activity [2221].
- p38 MAPK phosphorylates Nav1.8 in loop 1 (L1) of the channel, leading to increases in current density but does not alter gating properties [2222].
- A number of cAMP-dependent phosphorylation sites were also identified in Nav1.8: one in domain II between S3 and S4 and another in the interdomain II-III [860].
These phosphorylation events do not only change the activity and gating of the protein but also alter its expression. Often times, phosphorylation of the protein increases its trafficking to the cell membrane, in turn making the neuron more excitable. [2128]
Nav1.8 is subject to ubiquitination as it possesses a PY motif and was shown to be negatively regulated by Nedd4-2. This regulation was further demonstrated by experiments that over- and under-expressed Nedd4-2, leading to respective increase and decrease densities of Nav1.8 and neuron excitability. [2128]
Visual Representation of Nav1.8 Structure
Methodology for visual representation of structure available here
Like all voltage gated sodium channels, Nav1.8 is made up of a single protein comprised of 4 homologous domains (DI-DIV). Each domain is made up of 6 transmembrane subunits (S1-S6). S1-4 form the voltage sensing domain (VSD) whereas the S5-6 form the pore module (PM). The S4 subunit of each domain contains a series of positively charged residues. When membrane depolarization occurs, these charged residues cause the movement of the S4 subunit, inducing a conformational change in S5-S6, opening of the channel and allowing the entry of sodium ions into the cell. Soon after opening, rapid inactivation of Nav1.8 is instigated by the binding of the IFM motif, found in the loop between D3 and D4, to a hydrophobic receptor site next to the S6 in D4. This binding causes the shift of S6, allosterically closing the channel, thus deactivating the channel. Nav1.8 then returns to its resting state following the hyperpolarization of the cell membrane [2115].
The structure of human Nav1.8, in complex with its selective blocker A-803467, was resolved via cryo-electron microscopy, giving us a detailed insight to the specific structural features of Nav1.8. Structural resolution of the ion channel showed that only the extracellular interface of Domain I and Domain II-S5 have Nav1.8 unique residues that may be the main contributors to the channel’s unique kinetics (see Kinetics section) [2223].
Nav1.8’s resistance to tetrodotoxin is conferred by specific residues in the S5-S6 linker. Indeed, aromatic amino acid substitutions in this area make the channel TTX-sensitive. [2128] [860].
Nav1.8 predicted AlphaFold size
Methodology for AlphaFold size prediction and disclaimer are available here
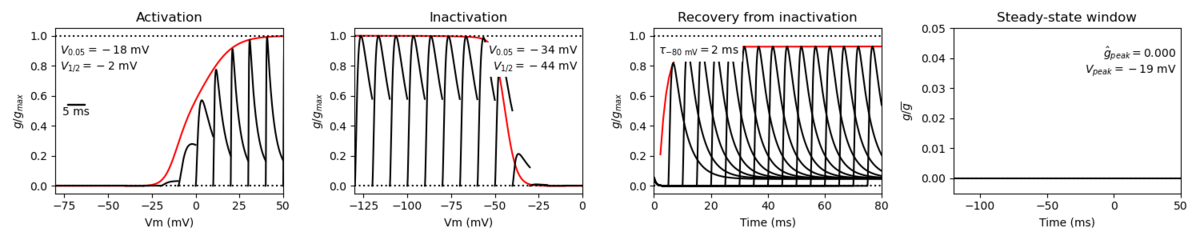
The electrophysiological activity of Nav1.8 is somewhat similar to that of other tetrodotoxin-resistant sodium channels [859].
However, the channel has distinctively slow gating kinetics. Nav1.8 exhibits slow-inactivating kinetics with a high threshold for activation, which generates a large inward current [1430]. These kinetics properties make Nav1.8 is a major contributor to the rising phase on the action potential [2224].
Single Channel Unitary Conductance
Single channel unitary conductance is determined experimentally.
For Nav1.8, single channel unitary conductance has yet to be determined experimentally.
Model
A single kinetic model for all human voltage-gated sodium channels (Balbi et al, 2017)
https://modeldb.science/230137
Species : Human | Gene: scn10a
Host cell: DRG neuron | Temperature: RT (to 25 C by Q10)
Formalism: Markov | States: C1, C2, O1, O2, I1, I2
Implementation: NEURON | Simulation: Nav18_a.mod
Tissue and Cellular
Nav1.8 is predominantly expressed in sensory neurons of the peripheral nervous system (PNS). More specifically, it is almost exclusively located in most peptidergic and non peptidergic small dorsal root ganglia (DRG). In situ hybridization using Nav1.8-specific oligonucleotide probes showed, that in the PNS, approximately 76% of the small neuronal cell population (400-1000 μm2) and 33% of the large cell population (1400-2000 μm2) were hybridized with probes for Nav1.8 [860]. It has also been detected, to a much lesser extent in the sciatic nerve, trigeminal ganglion, and nodose ganglia. [2225] [860] [2226].
Developmental
Nav1.8 is primarily expressed in adult cells. However, the channel’s expression starts in the embryo, at day 15, with expression then increasing to hit adult levels by postnatal day 7 [2124].
Nav1.8 is present at the unmyelinated nerve endings within sensory neurons [2227].
Action potential initiation
Given the channel’s kinetic properties, Nav1.8 is the major contributor to the upstroke of the action potential (AP) in sensory neurons, accounting for 80–90% of the inward current flow during the AP upstroke.
Nociception
Given its location, Nav1.8 is essential for AP firing in sensory neurons, allowing for the transmission of sensory information, particularly in sensitized nociceptors [1434]. Mouse models showed that Nav1.8-null mice had weak pain responses compared to their wild-type littermates, when subjected to stimulations that triggered a response from sensitized nociceptors [2228] [1436].
Indeed, Nav1.8’s role in nociception is further highlighted by its increased excitability to cold temperatures. Contrary to other sodium channels, whose activity decreases with lower temperatures, Nav1.8’s inactivation properties are cold-resistant, enabling the channel to fire at low temperatures and relay the information to the central nervous system. This property of Nav1.8 represents an important means of protection, in warm blooded animals, to help them detect and avoid tissue damaging at low temperatures [1430]
Pain & other channelopathies
However, as Nav1.8 is involved in nociception, deregulation of the channel often leads to issues with chronic pain and inflammation.
Several studies suggest that Nav1.8 plays an important role in inflammatory hyperexcitability as the channel’s expression is upregulated following certain noxious stimuli (intracolonic capsaicin or mustard oil). Nav1.8 knockout mice do not present the same response to these inflammatory compounds [1434] Intraplantar injection of complete Freund's adjuvant (CFA) experiments in rats also showed an increase in Nav1.8 expression and its accumulation at the sites of nerve injury in human patients with chronic neuropathic pain and chronic local hyperalgesia [2229]
Hyperexcitability of the channel is responsible for increased pain in conditions such as painful small fiber neuropathy (SFN). Two functional variants of Nav1.8, one that enhances ramp current and another shifting activation in a hyperpolarizing direction, render DRG neurons hyperexcitable in idiopathic SFN patients [2212] [2224]
Mutant Nav1.8 channels are also responsible for a number of other channelopathies:
Nav1.8 channels interact with various commercially available compounds such as menthol, lidocaine, tetracaine, vinpocetine, ambroxol, lamotrigine, mexilitine, veratridine, and A-803467, whereas very few animal toxins have been shown to be capable of reshaping Nav1.8 currents [1429]. For example, lidocaine, suppresses Na+ currents by binding not only to DIV–S6 but also to S6 of DI and DII, blocking the channels in a use-dependent (frequency-dependent) and voltage-dependent manner [856], [857]. Lidocaine enhanced current decrease in a frequency-dependent manner. Steady-state inactivation of Nav1.8 and Nav1.7 channels was also affected by lidocaine, Nav1.7 being the most sensitive. Only the steady-state activation of Nav1.8 was affected while the entry of both channels into slow inactivation was affected by lidocaine, Nav1.8 being affected to a larger degree. [217]
Beta subunits [220]
Nav1.8 is likely to be co-expressed with the b1, b2, and b3-subunits in C-fiber neurons from the DRG, and these subunits interact and modulate the channel.
b1-subunit alone, or in combination with other b-subunits, accelerates the time constant of inactivation and shifts the voltage-dependence of activation to more hyperpolarized potentials. Beta-1 also increases levels of Nav1.8 channel functional expression, either by increasing channel density at the oocyte membrane or by increasing individual channel conductance.
b3-subunit shifts the voltage-dependence of inactivation to more positive potentials and do not influence the time constant of current decay, steadystate activation or expression of Nav1.8. Co-expression of b1 and b3-subunits with Nav1.8 resulted in little change in availability compared to channel alone.
b2-subunit do not alter the kinetic properties or current amplitude of However, the co-expression of b1 and b2 shift the Nav1.8 channel availability to more depolarized potentials in comparison to when channels are expressed with either b1 or b2 alone.
Other proteins that interact with Nav1.8 [1431]
p11 directly binds only to NaV1.8 and translocate it to the plasma membrane.
PDZD2 and syntrophin-associated serine/threonine kinase (SASTK) are linked to the functional expression of NaV1.8 on the plasma membrane.
Contactin regulates the surface expression of Nav1.8.
CAP-1A binds to a conserved motif present in NaV1.8 linking voltage-gated sodium channels to clathrin, which is involved in coated vesicle assembly.
Tetrodotoxin
Nav1.8 is TTX resistant [1376]
- Known and predicted drug interactions with Nav1.8
- Known and predicted animal toxin interactions with Nav1.8
References
Chevrier P
et al.
Differential modulation of Nav1.7 and Nav1.8 peripheral nerve sodium channels by the local anesthetic lidocaine.
Br. J. Pharmacol.,
2004
Jun
, 142 (576-84).
Choi JS
et al.
Differential slow inactivation and use-dependent inhibition of Nav1.8 channels contribute to distinct firing properties in IB4+ and IB4- DRG neurons.
J. Neurophysiol.,
2007
Feb
, 97 (1258-65).
Browne LE
et al.
Functional and pharmacological properties of human and rat NaV1.8 channels.
Neuropharmacology,
2009
Apr
, 56 (905-14).
Vijayaragavan K
et al.
Role of auxiliary beta1-, beta2-, and beta3-subunits and their interaction with Na(v)1.8 voltage-gated sodium channel.
Biochem. Biophys. Res. Commun.,
2004
Jun
25
, 319 (531-40).
John VH
et al.
Heterologous expression and functional analysis of rat Nav1.8 (SNS) voltage-gated sodium channels in the dorsal root ganglion neuroblastoma cell line ND7-23.
Neuropharmacology,
2004
Mar
, 46 (425-38).
Rabert DK
et al.
A tetrodotoxin-resistant voltage-gated sodium channel from human dorsal root ganglia, hPN3/SCN10A.
Pain,
1998
Nov
, 78 (107-14).
Vijayaragavan K
et al.
Gating properties of Na(v)1.7 and Na(v)1.8 peripheral nerve sodium channels.
J. Neurosci.,
2001
Oct
15
, 21 (7909-18).
Ragsdale DS
et al.
Molecular determinants of state-dependent block of Na+ channels by local anesthetics.
Science,
1994
Sep
16
, 265 (1724-8).
Kretschmer T
et al.
Accumulation of PN1 and PN3 sodium channels in painful human neuroma-evidence from immunocytochemistry.
,
2002
Aug
, 144 (803-10; discussion 810).
Akopian AN
et al.
A tetrodotoxin-resistant voltage-gated sodium channel expressed by sensory neurons.
Nature,
1996
Jan
18
, 379 (257-62).
Sangameswaran L
et al.
Structure and function of a novel voltage-gated, tetrodotoxin-resistant sodium channel specific to sensory neurons.
J. Biol. Chem.,
1996
Mar
15
, 271 (5953-6).
Cummins TR
et al.
Downregulation of tetrodotoxin-resistant sodium currents and upregulation of a rapidly repriming tetrodotoxin-sensitive sodium current in small spinal sensory neurons after nerve injury.
J. Neurosci.,
1997
May
15
, 17 (3503-14).
Elliott AA
et al.
Characterization of TTX-sensitive and TTX-resistant sodium currents in small cells from adult rat dorsal root ganglia.
J. Physiol. (Lond.),
1993
Apr
, 463 (39-56).
Schild JH
et al.
Experimental and modeling study of Na+ current heterogeneity in rat nodose neurons and its impact on neuronal discharge.
J. Neurophysiol.,
1997
Dec
, 78 (3198-209).
Blair NT
et al.
Roles of tetrodotoxin (TTX)-sensitive Na+ current, TTX-resistant Na+ current, and Ca2+ current in the action potentials of nociceptive sensory neurons.
J. Neurosci.,
2002
Dec
1
, 22 (10277-90).
Renganathan M
et al.
Contribution of Na(v)1.8 sodium channels to action potential electrogenesis in DRG neurons.
J. Neurophysiol.,
2001
Aug
, 86 (629-40).
Djouhri L
et al.
The TTX-resistant sodium channel Nav1.8 (SNS/PN3): expression and correlation with membrane properties in rat nociceptive primary afferent neurons.
J. Physiol. (Lond.),
2003
Aug
1
, 550 (739-52).
Blair NT
et al.
Role of tetrodotoxin-resistant Na+ current slow inactivation in adaptation of action potential firing in small-diameter dorsal root ganglion neurons.
J. Neurosci.,
2003
Nov
12
, 23 (10338-50).
Nassar MA
et al.
Neuropathic pain develops normally in mice lacking both Na(v)1.7 and Na(v)1.8.
,
2005
, 1 (24).
Nassar MA
et al.
Nociceptor-specific gene deletion reveals a major role for Nav1.7 (PN1) in acute and inflammatory pain.
Proc. Natl. Acad. Sci. U.S.A.,
2004
Aug
24
, 101 (12706-11).
Gilchrist J
et al.
Animal toxins can alter the function of Nav1.8 and Nav1.9.
Toxins (Basel),
2012
Aug
, 4 (620-32).
Zimmermann K
et al.
Sensory neuron sodium channel Nav1.8 is essential for pain at low temperatures.
Nature,
2007
Jun
14
, 447 (855-8).
Liu M
et al.
The roles of sodium channels in nociception: implications for mechanisms of neuropathic pain.
Pain Med,
2011
Jul
, 12 Suppl 3 (S93-9).
Faber CG
et al.
Gain-of-function Nav1.8 mutations in painful neuropathy.
Proc. Natl. Acad. Sci. U.S.A.,
2012
Nov
20
, 109 (19444-9).
Hillsley K
et al.
Dissecting the role of sodium currents in visceral sensory neurons in a model of chronic hyperexcitability using Nav1.8 and Nav1.9 null mice.
J. Physiol. (Lond.),
2006
Oct
1
, 576 (257-67).
Abrahamsen B
et al.
The cell and molecular basis of mechanical, cold, and inflammatory pain.
Science,
2008
Aug
1
, 321 (702-5).
Akopian AN
et al.
The tetrodotoxin-resistant sodium channel SNS has a specialized function in pain pathways.
Nat. Neurosci.,
1999
Jun
, 2 (541-8).
Braz JM
et al.
Parallel "pain" pathways arise from subpopulations of primary afferent nociceptor.
Neuron,
2005
Sep
15
, 47 (787-93).
Black JA
et al.
Molecular identities of two tetrodotoxin-resistant sodium channels in corneal axons.
Exp. Eye Res.,
2002
Aug
, 75 (193-9).
Henry MA
et al.
Localization of the Nav1.8 sodium channel isoform at nodes of Ranvier in normal human radicular tooth pulp.
Neurosci. Lett.,
2005 May 20-27
, 380 (32-6).
Benn SC
et al.
Developmental expression of the TTX-resistant voltage-gated sodium channels Nav1.8 (SNS) and Nav1.9 (SNS2) in primary sensory neurons.
J. Neurosci.,
2001
Aug
15
, 21 (6077-85).
Laedermann CJ
et al.
Post-translational modifications of voltage-gated sodium channels in chronic pain syndromes.
Front Pharmacol, 2015, 6 (263).
Huang J
et al.
Small-fiber neuropathy Nav1.8 mutation shifts activation to hyperpolarized potentials and increases excitability of dorsal root ganglion neurons.
J. Neurosci.,
2013
Aug
28
, 33 (14087-97).
Widmark J
et al.
Differential evolution of voltage-gated sodium channels in tetrapods and teleost fishes.
Mol. Biol. Evol.,
2011
Jan
, 28 (859-71).
Fitzgerald EM
et al.
cAMP-dependent phosphorylation of the tetrodotoxin-resistant voltage-dependent sodium channel SNS.
J. Physiol. (Lond.),
1999
Apr
15
, 516 ( Pt 2) (433-46).
Hudmon A
et al.
Phosphorylation of sodium channel Na(v)1.8 by p38 mitogen-activated protein kinase increases current density in dorsal root ganglion neurons.
J. Neurosci.,
2008
Mar
19
, 28 (3190-201).
Huang X
et al.
Structural basis for high-voltage activation and subtype-specific inhibition of human Nav1.8.
Proc Natl Acad Sci U S A, 2022Jul26, 119 (e2208211119).
Xiao Y
et al.
Increased Resurgent Sodium Currents in Nav1.8 Contribute to Nociceptive Sensory Neuron Hyperexcitability Associated with Peripheral Neuropathies.
J Neurosci, 2019Feb20, 39 (1539-1550).
Zerres K
et al.
Spinal muscular atrophy--clinical and genetic correlations.
Neuromuscul Disord, 1997May, 7 (202-7).
Fukuyama M
et al.
Novel SCN10A variants associated with Brugada syndrome.
Europace, 2016Jun, 18 (905-11).
Brock JA
et al.
Tetrodotoxin-resistant impulses in single nociceptor nerve terminals in guinea-pig cornea.
J. Physiol. (Lond.),
1998
Oct
1
, 512 ( Pt 1) (211-7).
Laird JM
et al.
Deficits in visceral pain and referred hyperalgesia in Nav1.8 (SNS/PN3)-null mice.
J. Neurosci.,
2002
Oct
1
, 22 (8352-6).
Su YY
et al.
KIF5B promotes the forward transport and axonal function of the voltage-gated sodium channel Nav1.8.
J. Neurosci.,
2013
Nov
6
, 33 (17884-96).
Bengel P
et al.
Detrimental proarrhythmogenic interaction of Ca2+/calmodulin-dependent protein kinase II and NaV1.8 in heart failure.
Nat Commun, 2021Nov15, 12 (6586).
Cleary CM
et al.
Disordered breathing in a Pitt-Hopkins syndrome model involves Phox2b-expressing parafacial neurons and aberrant Nav1.8 expression.
Nat Commun, 2021Oct13, 12 (5962).
El-Battrawy I
et al.
A cellular model of Brugada syndrome with SCN10A variants using human-induced pluripotent stem cell-derived cardiomyocytes.
Europace, 2019Sep01, 21 (1410-1421).
Credits
To cite this page: [Contributors] Channelpedia https://channelpedia.epfl.ch/wikipages/127/ , accessed on 2024 May 04